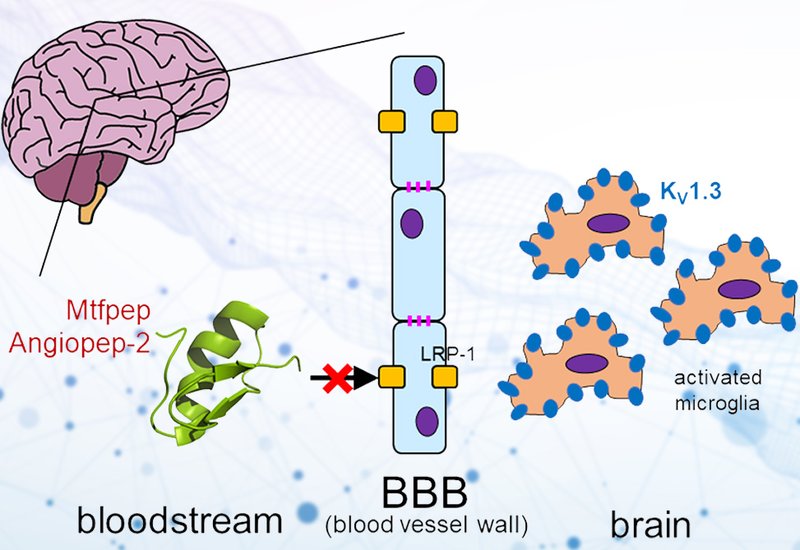
Researchers supervised by Professor Raymond S. Norton at Monash University, Australia, published in the International Journal of Peptide Research and Therapeutics, developed a modular click chemistry platform for conjugating blood-brain barrier shuttle peptides to a venom-derived potassium channel blocker, revealing that the shuttle peptides tested did not enhance brain delivery of the peptide cargo.
Neuroinflammation drives progression in Alzheimer's and Parkinson's disease, and the voltage-gated potassium channel KV1.3 has emerged as a compelling therapeutic target. This channel activates pro-inflammatory microglia in the brain, and its blockade reduces neuroinflammation in animal models. The most potent and selective KV1.3 blockers are disulfide-rich, venom-derived peptides that offer far greater selectivity than small-molecule inhibitors. Among these, HsTX1[R14A], a 34-residue scorpion toxin analogue with four disulfide bonds, blocks KV1.3 with an IC50 of approximately 45 pM and more than 2,000-fold selectivity over related channels. Peripheral administration of this peptide has improved cognition in mouse models of Alzheimer's disease and reduced neuroinflammatory markers in lipopolysaccharide-challenged mice. However, the blood-brain barrier largely excludes such structured peptides from the brain, limiting the concentrations achievable at the presumed site of action. BBB shuttle peptides offer a promising delivery strategy by targeting endogenous transport receptors on brain endothelial cells to promote receptor-mediated transcytosis. Two well-characterized shuttles, MTfpep derived from melanotransferrin and the synthetic peptide Angiopep-2, have previously enhanced brain uptake of antibodies, enzymes and nanoparticles by several fold in preclinical models.
The team conjugated both shuttle peptides to HsTX1[R14A] using copper-catalyzed azide-alkyne cycloaddition. They functionalized the peptide's N-terminus with azidolysine and separately equipped each shuttle with an alkyne handle via hexynoic acid coupling. This modular strategy overcame the poor synthetic yields and refolding failures encountered when attempting to produce the shuttle-peptide conjugates as single polypeptide chains. The click conjugation proceeded in 50 to 70 percent yield on the folded, disulfide-intact scaffold. NMR spectroscopy confirmed that both conjugates retained the native fold of HsTX1[R14A], and electrophysiology assays showed that MTf-HsTX1[R14A] and Ang2-HsTX1[R14A] blocked KV1.3 with IC50 values of 0.8 and 1.4 nM, respectively, representing only modest losses in potency consistent with other N-terminal modifications of this peptide.
Despite these encouraging biophysical results, neither conjugate demonstrated enhanced brain permeability. In two in vitro BBB models using primary mouse brain endothelial cells and an immortalized human brain endothelial cell line, MTf-HsTX1[R14A] showed no improvement over the unconjugated peptide, with both compounds transferring less than one percent across the cell layer over 120 minutes. Intravenous administration of MTf-HsTX1[R14A] to C57BL/6 mice at 4 mg/kg produced detectable plasma concentrations for up to two hours, but brain levels remained below the limit of detection at all time points. The Angiopep-2 conjugate could not be evaluated further due to insufficient analytical sensitivity in the LC-MS/MS assay and adverse effects observed at the tested dose in preliminary mouse experiments.
Although the shuttle peptides failed to deliver their cargo across the blood-brain barrier in this context, the click chemistry conjugation platform itself represents a significant methodological advance for the field. The approach enables modular assembly of disulfide-rich peptides with diverse targeting ligands while preserving both structural integrity and channel-blocking activity. The validated LC-MS/MS quantification methods for measuring peptide concentrations in plasma and brain tissue provide essential tools for future pharmacokinetic studies. These results also highlight an important cautionary finding: BBB shuttle peptides that succeed with larger cargo molecules like antibodies and enzymes do not necessarily translate to structured peptide payloads. Future efforts will explore alternative brain-targeting strategies using this same conjugation framework.