Collagen is the most abundant structural protein in the extracellular matrix and the mechanical backbone of most mammalian tissues. Materials built from fibrillar collagen find wide use in tissue engineering, drug delivery, and regenerative medicine, but natural collagen carries risks of immunogenicity and pathogen contamination, and its modification chemistry is constrained. Collagen-mimetic peptides, CMPs, offer a synthetic alternative: they can be efficiently prepared by SPPS and functionalized at will. The core challenge is that short CMPs form compact triple helices rather than the long, continuous filaments that give natural tropocollagen its structural strength. Existing strategies for elongating CMP assemblies, including disulfide-mediated "sticky ends" and electrostatic chain-register encoding, have yielded filaments only a few hundred nanometers long and require branched or multiply charged sequences that complicate synthesis and functionalization.
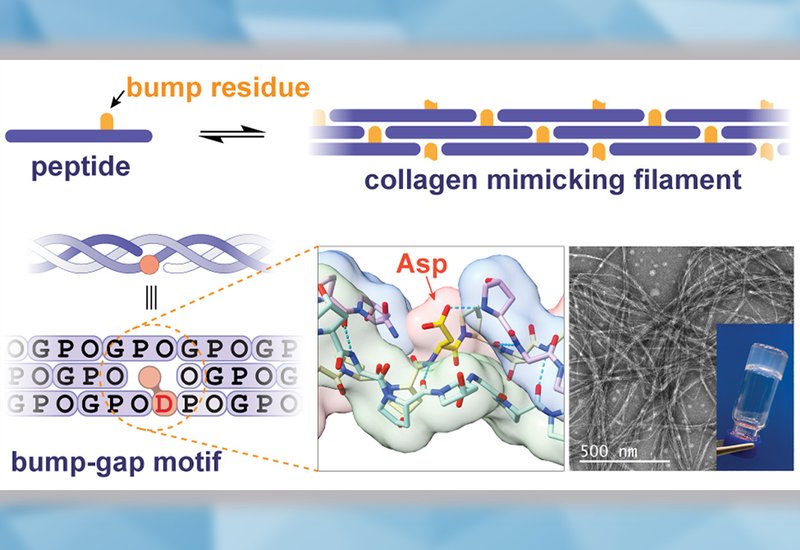
Researchers in the Chenoweth Group at the University of Pennsylvania, published in the Journal of the American Chemical Society, designed a new class of filament-forming CMPs that encodes chain-register specificity through steric rather than electrostatic interactions. The strategy places a single side-chain "bump" residue on one peptide strand so that it fits precisely into the structural "gap" created at the junction between successive triple-helical repeating units. Because the tightly packed collagen triple helix strongly destabilizes any non-glycine residue at an interior position, the bump residue is tolerated only in the specific staggered register that aligns it with the gap, automatically blocking all competing registers without requiring stabilization of the target register directly.
The team systematically varied peptide length, aza-glycine, azGly, content, bump residue identity, and gap geometry to arrive at peptide 4, a 28-residue sequence bearing an aspartic acid bump near its C-terminus. AzGly substitutions, which stabilize the collagen triple helix through additional hydrogen bonding and conformational preorganization, were included but deliberately reduced relative to earlier variants to suppress competing homotrimer formation. Circular dichroism, CD, analysis confirmed pH-dependent triple-helix formation: at acidic pH the aspartate side chain is protonated and the assembly melts at 25 °C, whereas buffering to neutral pH allows a salt bridge between the Asp bump and the N-terminal hydroxyproline of the gap to form, raising the melting temperature to 40 °C and driving supramolecular polymerization. Size-exclusion chromatography with multiangle light scattering confirmed multimeric aggregates at low temperature, and concentration-dependent CD measurements provided evidence for both individual filaments and filament bundles.
Transmission electron microscopy, cryogenic electron microscopy, cryo-EM, and atomic force microscopy, AFM, images show that peptide 4 self-assembles into rigid, micrometer-length filaments with a diameter of 1.7 nm, consistent with the 1.5 nm diameter of a canonical collagen triple helix. After 7 days of growth at 4 °C the filaments bundle into thicker assemblies, with width increasing from 4 to 8 nm and persistence length rising from 0.8 to 1.9 μm. Small-angle X-ray scattering confirmed an interhelix spacing of 1.2 nm within the nanofibers. At 2 mM concentration and neutral pH, peptide 4 gels within 1 minute of buffering. Oscillatory shear rheology shows storage moduli of 1.6 × 103 Pa, 1.0 × 104 Pa, and 3.6 × 104 Pa at 0.5%, 1.0%, and 2.0% w/v, respectively, placing the 0.5% hydrogel at a storage modulus 3-fold above bovine collagen and 160-fold above Matrigel under matched conditions. The gel transitions to a sol at 5% strain, a shear-thinning behavior the authors attribute to shear-induced fragmentation of the supramolecular filaments; bovine collagen remains gelled under 100% strain, and Matrigel shows no shear-thinning. Systematic mutagenesis confirmed the design logic: replacing the Asp bump with Asn, His, Cys, Phe, or Tyr abolished gelation entirely, while the Glu bump gave only weak gelation, supporting the necessity of the salt-bridge interaction. Altering gap size, bump position, peptide length, or terminal capping equally disrupted assembly.
The bump-gap strategy introduces a chemically minimal modification, a single side-chain substitution at one glycine position per repeating unit, that encodes chain-register specificity by destabilizing all undesired registers rather than stabilizing the preferred one. Because the single steric-bump residue is sufficient to impose the register, the rest of the peptide sequence remains largely unconstrained and can be readily varied or functionalized. The pH-triggered gelation and pronounced shear-thinning properties suggest utility as an injectable, high-stiffness scaffold. More broadly, the principle that periodic steric complementarity can drive controlled supramolecular polymerization of short peptides into long, defined filaments opens a design route for collagen-mimetic hydrogels with tunable mechanical and protein-binding properties across a range of bioengineering applications.