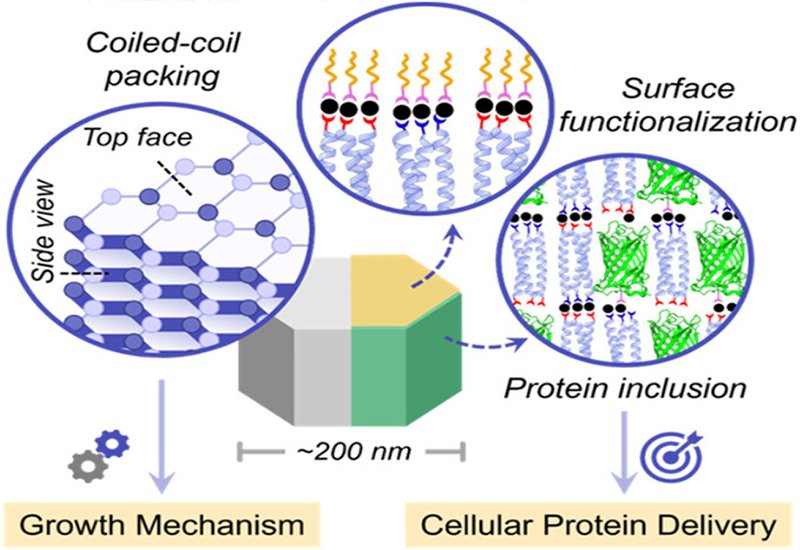
Coiled-coil peptides are prized architectural units in biomaterials design, capable of self-organizing into fibers, tubes, cages, and crystals across a wide range of length scales. Yet crystalline coiled-coil assemblies have mostly been explored at the micron scale, where their size precludes cellular uptake. Shrinking those architectures to the nanoscale while preserving their capacity for ordered protein inclusion and adding active cell-entry machinery would unlock a new class of intracellular protein delivery vehicle. That intersection of structural precision and biological function has remained largely unexplored.
Researchers in the Chmielewski Group at Purdue University, published in Chemistry of Materials, built on their earlier work with Zn(II)-promoted microcrystals of the trimeric coiled-coil peptide GCN4-p2L to engineer a nanoscale analogue. Switching to Ni(II) and carefully tuning buffer pH and metal stoichiometry yielded well-defined nanocrystals averaging approximately 180 nm in height and 165 nm in width. Negative-stain transmission electron microscopy, TEM, and small- and wide-angle X-ray scattering, SWAXS, resolved coiled-coil packing within the crystals. Temperature- and salt-dependent assembly experiments then probed the thermodynamic and kinetic forces governing crystal growth, and metal-ligand chemistry loaded protein cargo and decorated crystal surfaces with a cell-penetrating peptide.
TEM imaging of the nanocrystals revealed periodic horizontal banding along their length, with stacks of three layers within approximately 12 nm matching the known ~4 nm length of the GCN4-p2L coiled-coil. A top-face view showed open hexagonal packing in which each coiled-coil at a vertex contacts three neighbors, mirroring the arrangement seen in the Zn(II) microcrystal X-ray structure. SWAXS confirmed d-spacings of 4.35, 2.57, 1.94, and 0.97 nm and a unit cell lattice parameter of 3.88 nm, fully consistent with the open hexagonal lattice. Temperature-dependent growth experiments illuminated the growth mechanism: assembly at 4 °C yielded wide, disc-like crystals attributed to kinetically favored electrostatic interactions driving radial growth, while assembly at 37 °C produced tall, thin crystals reflecting thermodynamically preferred metal-ligand interactions that favor axial elongation. Addition of sodium halide salts at 20 mM enhanced ordering on the hexagonal top face without disturbing the banding pattern.
Taking advantage of Ni(II)-mediated metal-ligand coordination, the team co-assembled His-tagged enhanced green fluorescent protein, eGFP, within the growing nanocrystals. Quantification revealed approximately 750 His-tagged eGFP molecules per nanocrystal at a coiled-coil-to-protein ratio of 200:1, and confocal microscopy confirmed that the incorporated protein remained folded and fluorescent. Crystal morphology was preserved throughout. For cellular delivery, nanocrystals were subsequently incubated with additional Ni(II) and then with the His6-TAT peptide, loading approximately 2,700 TAT molecules per crystal onto surface-exposed metal-binding sites. Flow cytometry showed that unmodified eGFP-loaded nanocrystals entered HeLa cells above background, but TAT-decorated nanocrystals produced a threefold increase in cellular fluorescence. Confocal colocalization studies revealed that unmodified nanocrystals accumulated in endosomes, whereas TAT-functionalized nanocrystals escaped to the cytosol, a shift consistent with the known membrane-disrupting activity of TAT at high surface density. No cytotoxicity was observed for any nanocrystal formulation or for free Ni(II) at concentrations up to 400 μM.
These results establish crystalline coiled-coil nanostructures as a modular platform for intracellular protein delivery in which cargo loading and surface functionalization are both controlled by metal-ligand chemistry without disrupting the underlying crystal architecture. The ability to tune crystal morphology through temperature and ionic conditions, load native folded proteins at high density, and redirect subcellular trafficking via surface-displayed cell-penetrating peptides points toward a versatile design logic. Future work may exploit the observed degradation of nanocrystals in cell culture media as a programmable release mechanism, while the modular metal-coordination approach could be extended to diverse His-tagged therapeutic proteins and alternative targeting ligands.