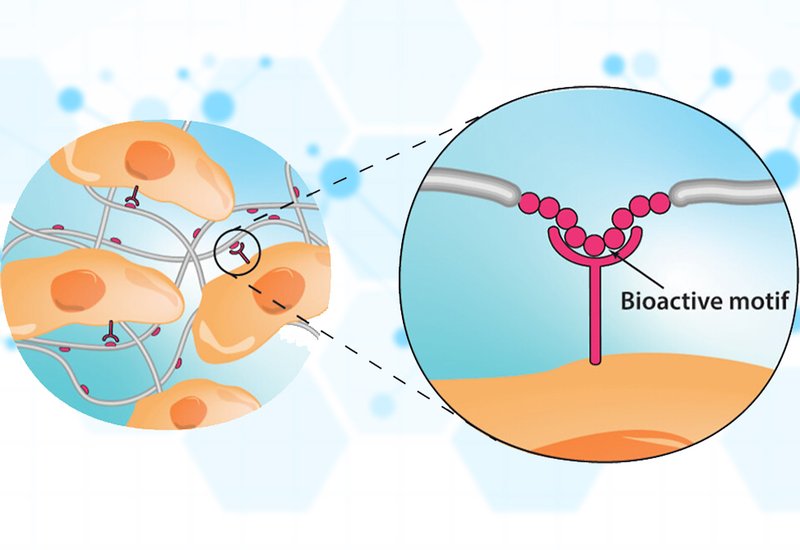
Researchers in the López-Silva Group at the Massachusetts Institute of Technology, published in Biochemistry, have in a recent review, outlined emerging design strategies for integrating bioactive peptides into advanced biomaterials that better mimic the dynamic complexity of natural tissue environments. While peptide-functionalized materials have long been used to control cell behavior, traditional approaches often rely on simply adding multiple cues without fully considering the biochemical context in which peptides operate. This review shifts the focus toward mechanistic design principles that account for synergistic interactions, structural conformation, peptide presentation, stimulus responsiveness, and intrinsic material properties.
A central theme is the importance of synergistic peptide motifs. In fibronectin, for example, the integrin-binding RGD tripeptide cooperates with the PHSRN sequence to selectively engage α5β1 integrins when presented at an optimal 30 to 40 Å separation. Incorporating both motifs into PEG hydrogels has enabled more accurate in vitro models of pancreatic cancer, enhanced bone regeneration on titanium implants, and improved the biocompatibility of decellularized heart valves. Similar synergy is exploited in immunoengineering: costimulatory antibodies on microcapsules activate T cells, while fusion peptides targeting both VEGFR-2 and neuropilin-1 enhance blood–brain barrier penetration for glioma gene therapy. Self-assembling Q11 nanofibers decorated with vaccine epitopes and adjuvants induce robust immune responses by presenting antigens at the 25 to 30 nm spacing optimal for B-cell receptor activation.
Structural conformation is equally critical. Cyclic RGD derivatives, which constrain the peptide into a kinked conformation, bind selectively to αvβ3 and αvβ5 integrins and consistently outperform linear RGD across diverse hydrogel and surface platforms. Cyclo-RGDfC functionalized gellan-gum gels promoted endothelial vasculogenesis without exogenous growth factors, and cyclo-RGDfK conjugated to alginate restored a healthy phenotype in degenerative nucleus pulposus cells. These observations align with broader principles in peptide therapeutics, where cyclization and stapling enhance potency and stability.
Peptide presentation, including linker chemistry, density, and spatial arrangement, profoundly affects bioactivity. Oligoproline linkers of at least three residues optimally displayed RGD on surfaces for cell adhesion, while longer PEG linkers improved PD-L1 chimera engagement with T cell receptors. Optimal peptide density varies with material context: 15% GDNF-mimetic peptide in self-assembling nanofibers supported neuron viability, whereas 10% and 33% formulations did not. Interpenetrating networks combining rigid PEG-RGD and mobile self-assembling peptide-RGD allowed dynamic focal adhesion formation, recapitulating the ligand mobility found in native extracellular matrix.
Stimulus-responsive designs introduce spatiotemporal control. Boolean logic-gated protease-labile tethers release proteins only when specific enzymatic inputs are met, enabling programmable bioactivity. Cryptic peptide sequences masked as nonfunctional depsipeptides rearrange into active YIGSR cell-adhesive motifs upon enzymatic cleavage. Light-responsive LOV2 protein switches control peptide display on demand with blue light, and multilayer hydrogels exploit pH-dependent degradation to sequentially release anti-inflammatory drugs and angiogenic growth factors during wound healing. These approaches translate molecular responsiveness into macroscopic tissue-level dynamics.
Intrinsic material properties also modulate peptide function. Protein adsorption onto charged or functionalized surfaces can drive cell responses independent of conjugated peptides. Positively charged self-assembling gels induce rapid neutrophil infiltration and extracellular trap formation, while negatively charged gels elicit macrophage-driven responses. Lysine-rich peptide scaffolds promote angiogenesis and innervation through acute inflammation, sometimes overshadowing the effects of tethered mimetic peptides. Understanding these material-driven interactions is essential to ensure that bioactive peptides perform as intended.