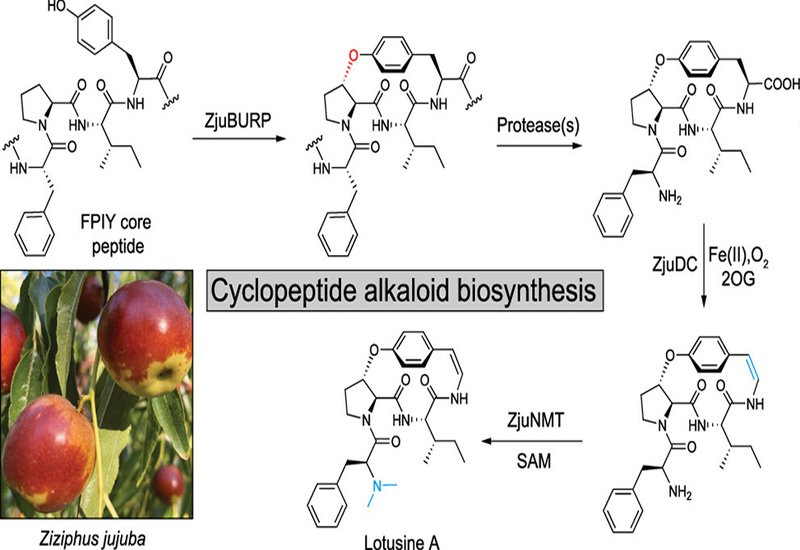
Cyclopeptide alkaloids, CPAs, are a family of plant-derived macrocyclic peptides found predominantly in buckthorn family plants, defined by a tyrosine-derived ether cross-link that forms strained 13- to 15-membered ring systems. Members of this class carry a range of medicinal activities, including analgesic effects from adouetine X, anxiolytic activity from sanjoinine A, and antiviral properties from the jubanines. Despite decades of interest and considerable synthetic effort, no biosynthetic pathway for any classical CPA had been reported, leaving the molecular machinery behind these scaffolds entirely unknown and limiting access to analogs for drug development.
Researchers in the Kersten Group at the University of Michigan, published in J. Am. Chem. Soc., identified candidate biosynthetic genes in the genome of the Chinese date tree, Ziziphus jujuba, and expressed them transiently in Nicotiana benthamiana to reconstitute pathway steps in planta. Differential gene expression analysis by RNA sequencing, RNA-seq, of young jujube stem and leaf tissues guided the discovery of enzyme candidates coexpressed with a known burpitide cyclase gene. Purified enzymes were then subjected to in vitro reconstitution assays using cyclic peptide intermediates produced from transgenic tobacco at milligram scale.
The team characterized three enzymes that together complete the biosynthesis of 14-membered CPAs. The split burpitide cyclase ZjuBURP macrocyclizes a ribosomal precursor peptide by forming an ether bond between a C-terminal tyrosine phenol and the β-carbon of a proline or leucine residue. Fusing ZjuBURP with its truncated precursor peptide increased cyclic product yields 2.9-fold in transgenic tobacco, enabling reproducible milligram-scale production of the key intermediate cyFPIY. The second enzyme, ZjuDC, is a nonheme iron- and 2-oxoglutarate-dependent dioxygenase that catalyzes oxidative decarboxylation of the C-terminal residue, installing the characteristic hydroxystyrylamine enamide of classical CPAs. Confirmed by in vitro reconstitution with Fe(II) and 2-oxoglutarate, with succinate detected as a coproduct, this reaction represents the first example of a 2-oxoglutarate-dependent nonheme iron enzyme performing peptide oxidative decarboxylation. Kinetic characterization of ZjuDC gave a Km of 3.51 μM and a kcat of 0.67 min-1. The third enzyme, ZjuNMT, is a SAM-dependent methyltransferase that installs α-N,N-dimethylation at the peptide N-terminus. Together, ZjuDC and ZjuNMT converted cyclic peptide intermediates derived from SkrBURP, a clubmoss burpitide cyclase, into the analgesic adouetine X and the anxiolytic sanjoinine A both in transgenic tobacco and in one-pot in vitro reactions confirmed against authentic standards. Systematic diversification by coexpressing SkrBURP constructs carrying all proteinogenic amino acid variants at variable core positions, then applying ZjuDC and ZjuNMT, generated 43% of possible analogs across three core peptide series in planta.
The elucidated pathway reduces what can require up to 20 synthetic steps into a five-enzyme biosynthetic sequence well suited to metabolic engineering and chemoenzymatic synthesis. ZjuDC expands the catalytic repertoire of the nonheme iron and 2-oxoglutarate enzyme superfamily to include peptide desaturation via oxidative decarboxylation, a reaction type with no prior precedent in this enzyme class. The broad substrate flexibility of both ZjuDC and ZjuNMT, demonstrated across more than 40 cyclic peptide variants, positions them as versatile biocatalysts for generating structurally diverse CPAs and potentially other ribosomally synthesized and posttranslationally modified peptide, RiPP, classes, opening a scalable route to bioactive macrocycles for medicinal chemistry and drug development.