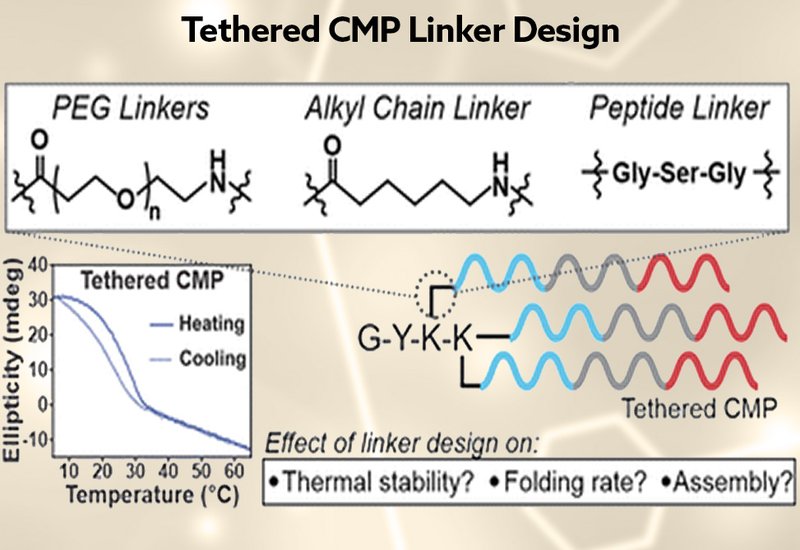
Collagen-mimetic peptides, CMPs, are synthetic tools for probing the folding rules of the collagen triple helix, and researchers have long sought ways to stabilize them beyond their naturally modest thermal stabilities. Covalent tethering to trivalent scaffolds addresses two chronic liabilities of untethered CMPs: concentration-dependent folding and sluggish refolding kinetics that can demand days or weeks to reach equilibrium. Yet despite the steady accumulation of tethered CMP designs in the literature, one design variable remains almost entirely unexplored: the linker that bridges each peptide strand to its scaffold. Linker length and chemistry set the conformational entropy cost of folding, offering a potential sequence-neutral knob for tuning triple-helix stability.
Researchers in the Merg Group at the University of California, Merced, published in RSC Chemical Biology, synthesized six tethered CMPs built on a charge-segregated triblock sequence, CMP322, known to direct triple helices into 2D nanosheets. Using a branched lysine strategy with orthogonal ivDde protecting-group chemistry and standard Fmoc solid-phase peptide synthesis, they prepared three PEGylated variants, CTH-PEG2, CTH-PEG4, and CTH-PEG6, alongside a hydrophobic aminohexanoic acid linker, CTH-HEX, a Gly-Ser-Gly peptide linker, CTH-GSG, and a no-linker control, CTH, with strands attached directly to the ε-amino groups of lysine. Circular dichroism, CD, with thermal denaturation, transmission electron microscopy, TEM, and atomic force microscopy, AFM, together characterized stability, folding kinetics, and nanostructure morphology.
Every construct except CTH-PEG6 assembled into TEM-detectable nanosheets and nanoribbons. AFM confirmed monolayer thicknesses of 5.8 ± 2.0 nm, matching the 6.0 nm length expected for a 21-residue triple helix at 0.286 nm rise per residue. The no-linker construct CTH proved the most thermally stable, with a melting temperature of 29.0 ± 0.5 °C, while each additional PEG unit eroded stability: CTH-PEG2 melted at 21.4 ± 0.3 °C, CTH-PEG4 at 18.0 ± 0.04 °C, and CTH-PEG6 at only 16.4 ± 0.3 °C, too low to support detectable assembly. The trend confirms that longer, more flexible linkers impose a greater entropic penalty on folding without compensating enthalpic gain from the unchanged CMP sequence. Crucially, the result overturns the common assumption that an extended linker, such as aminohexanoic acid, is needed to accommodate triple-helix geometry: the lysine side chain alone provides sufficient flexibility for the most stable constructs in the series.
Refolding hysteresis measurements, the first reported for end-capped CMPs, revealed that longer PEG linkers also slow fold recovery. ΔT values of 3.2 °C, 6.0 °C, and 7.1 °C for CTH-PEG2, CTH-PEG4, and CTH-PEG6, respectively, indicate progressively higher kinetic barriers to refolding. Linker composition introduced subtler effects: CTH-GSG melted at 24.0 °C and CTH-HEX at 22.2 °C, both slightly outperforming CTH-PEG2, likely because their lower conformational freedom reduces the entropic folding cost relative to a PEG chain of comparable contour length. The team further demonstrated that CTH assembles into nanoribbons and nanosheets at 50 µM, fourfold below the standard working concentration, confirming that covalent tethering renders assembly genuinely concentration-independent.
These findings reframe linker design as a practical, sequence-neutral strategy for dialing in the thermal stability and folding kinetics of tethered CMPs without touching the functional peptide domain. For researchers building collagen-mimetic biomaterials, the implication is direct: shorter or absent linkers preserve nanosheet formation while maximizing triple-helix stability, whereas longer linkers offer a deliberate route to reduced stability when that is the goal. The work also provides the first hysteresis benchmarks for this scaffold class, offering a quantitative basis for comparing future linker designs. Broader validation across other tethering strategies and CMP sequences remains open, but the framework gives the field a clear starting point.