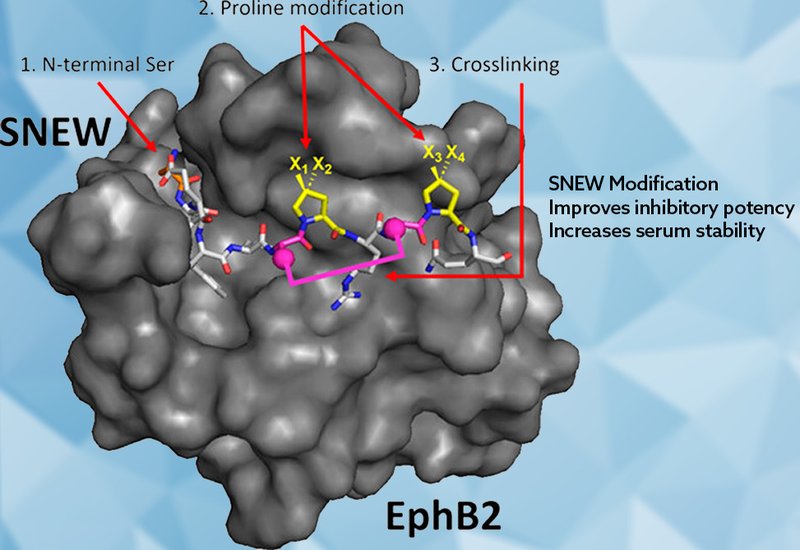
Ephrin receptors, Eph receptors, constitute the largest family of receptor tyrosine kinases in humans, with 14 members divided into A and B subclasses. Their ligand-binding surfaces are highly conserved, making receptor-selective inhibition difficult by conventional small-molecule approaches. Yet peptide phage display has repeatedly cracked this problem, yielding linear and cyclic peptides that bind individual receptors with high specificity. For the EphA2, EphA4, and EphB4 receptors, those phage-display hits have been refined to nanomolar potency. The EphB2-targeting peptide SNEW, SNEWIQPRLPQH, identified years ago with only modest inhibitory activity, has attracted far less optimization effort, despite EphB2 upregulation in colorectal cancer, Barrett’s esophageal neoplasia, and melanoma.
Researchers in the Sawyer Lab at Fordham University, published in Biochemistry, pursued two parallel modification strategies to improve SNEW’s inhibitory potency and serum stability without sacrificing its selectivity for EphB2. Starting from a truncated variant, SNEWIQPRLPQ, the team replaced the N-terminal serine with a panel of cyclic α-amino acids and separately replaced each of the two proline residues with conformationally biased proline analogs, including 4,4-difluoroproline, dFP. They then synthesized doubly and triply modified variants combining N-terminal substitutions, dFP at Pro7, and a previously validated 2,7-dimethylnaphthyl cross-link. Inhibitory potency was quantified via competitive enzyme-linked immunosorbent assay measuring blockade of the EphB2–ephrin B2 interaction, and serum stability was tracked by analytical HPLC after incubation in 50% human serum.
The most informative results emerged from the N-terminal series. Replacing L-serine with S-morpholine-3-carboxylic acid, which covalently links the N-terminal amine to the side-chain oxygen through an ethylene bridge, yielded a variant with an IC50 of 1.7 µM, nearly a 10-fold gain over the parent peptide’s IC50 of 15 µM. Structural analysis of the SNEW–EphB2 crystal complex suggested that the morpholine ring promotes a gauche(+) side-chain conformation, enabling new interactions with specific EphB2 residues, including packing contacts with Val164 and a hydrogen bond to Gln157 that compensate for rearranged interactions with Asp104 and Lys166. Related saturated six-membered-ring variants, including an L-homoproline analog and a thiomorpholine analog, matched this potency gain with IC50 values of 3.0 and 1.6 µM, respectively. Introducing unsaturation, removing the charge on the ring nitrogen, or repositioning the ring heteroatoms consistently eroded potency, confirming that a saturated, six-membered ring bearing a protonatable nitrogen at the N-terminus is the key pharmacophoric feature.
Among proline modifications, dFP substitution at Pro7 gave an IC50 of 5.5 µM, a roughly 3-fold potency gain over the parent and the best result in the proline series. The primary benefit of dFP proved to be more relevant to metabolism rather than potency: variants carrying dFP at Pro7, including N-terminally modified analogs, all showed slower serum degradation than their non-fluorinated counterparts. The half-life of the unmodified parent peptide and N-terminally modified variants was approximately 48 hours in 50% human serum, whereas dFP incorporation doubled that half-life to approximately 96 hours. Doubly and triply modified variants combining N-terminal cyclic residues, dFP, and the naphthyl cross-link showed no synergistic potency gain beyond the individual contributions, but they retained full EphB2 selectivity when tested against EphB1, EphB3, EphB4, and EphB6 at 50 µM.
These findings offer two practical handles for SNEW-based drug development and, more broadly, for peptide optimization campaigns. N-terminal serine-to-cyclic-amino-acid substitution improved potency by nearly an order of magnitude and may generalize to other phage-display-derived peptides whose N-terminal serine, threonine, or cysteine anchors a polar interaction network at a receptor interface. The dFP substitution at a central proline provides a serum-stabilization strategy that operates independently of conformational preferences and is compatible with concurrent potency-enhancing modifications. Together, the most potent and stable SNEW variants reported here are candidates for advancing toward cell-based and in vivo studies of EphB2 signaling in EphB2-upregulated cancers.