Obesity and metabolic syndrome remain formidable clinical challenges despite impressive advances in incretin-based therapies. GLP-1 agonists like semaglutide deliver substantial weight loss and glycemic control, yet they fall short of replicating the multifaceted benefits of bariatric surgery, particularly in correcting lipid abnormalities that drive residual cardiovascular risk. GLP-1 and GIP coagonists such as tirzepatide push the envelope further, but significant gaps persist in lipid lowering and maximal weight reduction. Fibroblast growth factor 21, FGF21, offers a complementary mechanism with potent effects on triglycerides, cholesterol, and hepatic fat without the gastrointestinal burden of incretins, suggesting a path to more comprehensive metabolic correction.
Researchers in the DiMarchi Group at Novo Nordisk Research Center Indianapolis, published in Biochemistry, engineered a first-in-class triple agonist that simultaneously activates FGF21, GLP-1, and GIP receptors. They began by optimizing native FGF21 through strategic N- and C-terminal modifications, introducing stabilizing disulfide bridges and mutations that enhance potency at FGFR1 while minimizing aggregation. The resulting Protein 5 displayed tenfold greater potency than native FGF21 in human cells and superior weight loss in diet-induced obese, DIO, mice. To extend duration of action, the team conjugated either a C18 fatty diacid or tetrazole to the N-terminus, yielding lipidated constructs that retained high potency in both albumin-bound and unbound states and demonstrated protracted pharmacokinetics suitable for reduced dosing frequency.
The lipidated FGF21 analogs achieved remarkable efficacy in DIO mice, with daily administration of 3 nanomoles per kilogram producing approximately 20 percent body weight loss within seven days. Extending the dosing interval to every third day maintained robust weight reduction, confirming sustained biological action. When compared head-to-head with a semaglutide surrogate at equivalent molar doses over 28 days, the optimized FGF21 analog surpassed the GLP-1 agonist by day 11 and ultimately achieved 43 percent absolute body weight reduction versus 30 percent for the incretin. Combination studies revealed additive effects: coadministration of Protein 5 with a GLP-1 and GIP coagonist peptide produced 18 percent weight loss in one week, significantly exceeding either agent alone. This synergy validated the rationale for integrating FGF21 pharmacology with incretin signaling in a single molecule.
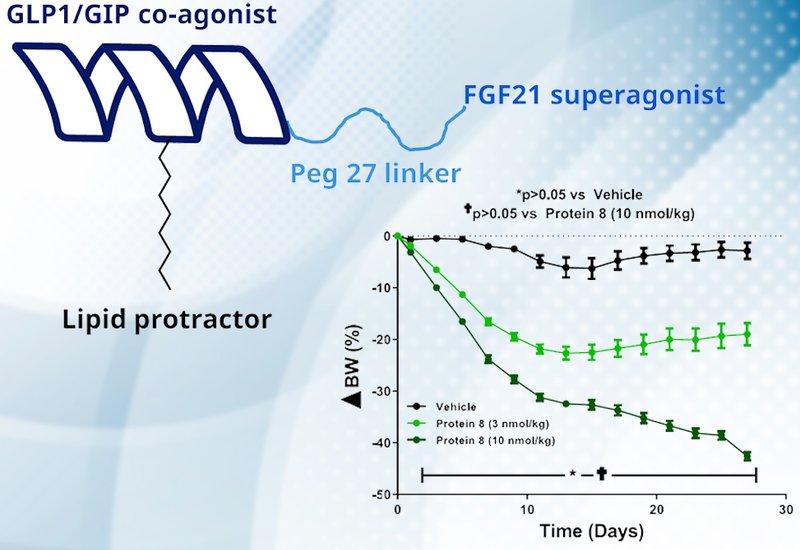
The researchers then synthesized Protein 8, a unimolecular triagonist, by conjugating a protracted GLP-1 and GIP coagonist peptide to Protein 5 via a polyethylene glycol spacer and chemoselective thioether ligation. The triagonist displayed balanced subnanomolar potency at all three human receptors in albumin-bound conditions, with picomolar incretin activity and low nanomolar FGF21 activity. Mechanistic validation used site-specific knockouts: silencing FGF21 activity by a D163A mutation or ablating incretin activity by N-terminal truncation each reduced efficacy relative to the intact triagonist, confirming dual pharmacology. In a 27-day study, daily administration of 10 nanomoles per kilogram of Protein 8 normalized body weight in DIO mice, achieving 43 percent reduction driven predominantly by fat mass loss with preservation of lean tissue. Glucose tolerance improved markedly, and the triagonist outperformed both selective FGF21 and incretin knockouts in reducing body weight and food intake.
The findings establish the FGF21, GLP-1, and GIP triagonist as a compelling candidate for next-generation metabolic therapy. By combining the lipid-lowering and energy-expenditure benefits of FGF21 with the appetite suppression and glycemic control of dual incretin agonism, Protein 8 addresses gaps left by current treatments and moves closer to pharmacologically replicating bariatric surgery. The convergent semisynthetic approach offers a blueprint for engineering balanced multiagonists with extended duration of action, and the translational promise warrants clinical evaluation in patients with obesity and metabolic syndrome.